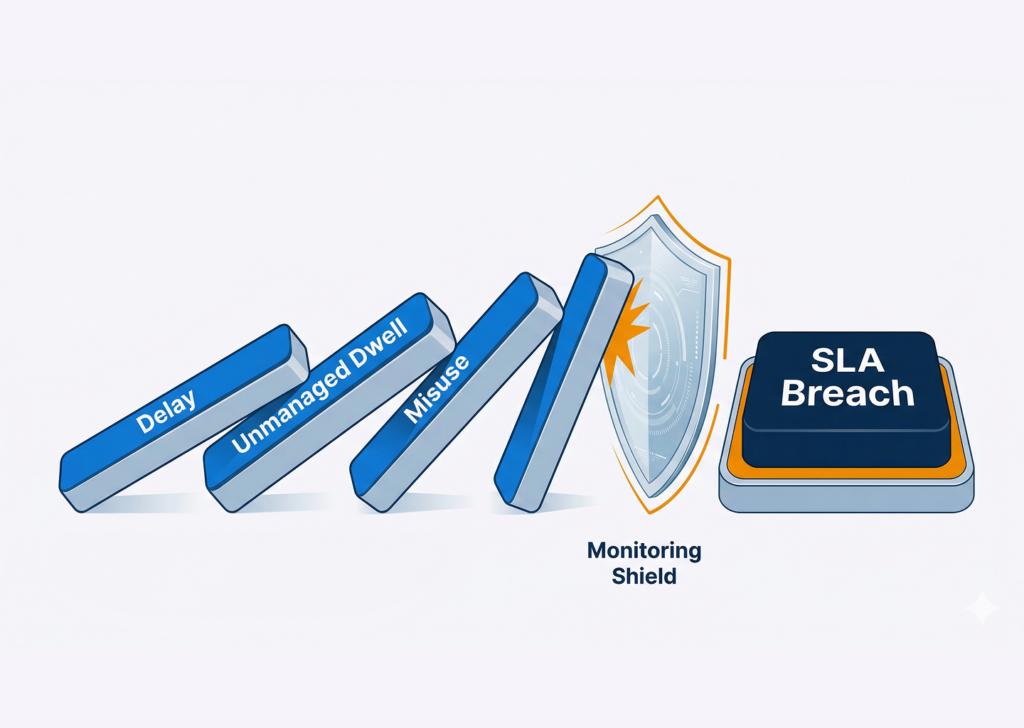
In pharma logistics, an SLA miss is rarely just a timing problem. It is often the visible result of deeper issues across the pharma supply chain: missing handoff records, delayed exception detection, poor condition visibility, and asset data that never reaches the teams responsible for acting on it.
In a sector where temperature, traceability, and documented movement matter as much as speed, that gap gets expensive fast.
WHO guidance for time- and temperature-sensitive pharmaceutical products and EU GDP principles both point to the same operational reality: storage, transport, and distribution controls cannot be treated as loose back-office processes.
They need to hold under real movement, real delays, and real handoffs.
This is where automated asset tracking starts to matter far beyond simple location visibility. In pharma logistics, tracking has to support condition assurance, recorded custody, and a usable history of movement that stands up when service, compliance, or product integrity is questioned.
That matters even more as regulations like DSCSA continue pushing the industry toward stronger electronic traceability across the pharmaceutical supply chain. A late shipment is one issue. A late shipment with incomplete records is a bigger one.
AI can help fix this, but not in the vague, fashionable way the market often talks about it. Its value is more practical. AI can help connect event streams, detect anomalies early, flag likely SLA risk before a breach happens, and surface which shipments, assets, or lanes need action first.
That fits the exact direction SensaTrak already uses across its platform language: contextual intelligence, predictive risk detection, automated SLA checks, and historical records that support audits and operations together.
McKinsey’s work on AI-enabled distribution control towers and Gartner’s work on digitally informed decision-making both reinforce the same idea: smarter logistics performance comes from earlier signals and faster action, not just more dashboards.
This blog will look at how AI can help reduce logistics SLA failures in pharma logistics, where traditional tracking falls short, and why automated asset tracking combined with condition monitoring and operational context is becoming necessary for a more resilient pharma supply chain.
Why are pharma logistics SLA issues hard to catch early?
These SLA issues in pharma logistics don’t get noticed very easily.
They begin earlier, during movement, handoffs, and storage conditions, but remain invisible until the outcome is already affected.
A shipment gets delayed, but the delay actually began at a prior node.
A temperature excursion occurs, but the risk built-up during transit.
A delivery misses its SLA, but the signal was present much earlier.
This is the core problem in the pharma supply chain. Most systems do not capture or connect events as they happen. They record outcomes after the fact.
WHO guidance for time- and temperature-sensitive pharmaceutical products emphasizes that storage and transport conditions must be maintained and monitored continuously, and not checked occasionally. EU GDP guidelines reinforce the need for documented control across the entire distribution process.
But as you know, the operational reality looks very different.
Handoffs are often manual.
Condition data is not continuous.
Exception alerts come late.
Records exist, but not in a form that supports action.
So, the SLAs appear to “fail” at delivery, while the actual failure occurred somewhere in transit or way before the delivery actually started, without being detected or acted upon.
That is why pharma logistics SLA issues are hard to catch early. The system is not designed to surface risk in time.
Why conventional tracking still leaves operational gaps?
Most tracking systems answer one some narrow questions: where is the asset? for how long the asset has been there? What route has the asset taken?
In pharma logistics, these factors are useful, but they don’t answer the operational questions completely.
Location, dwell time and route alone do not explain product condition, handling quality, dwell time, route deviation, or whether the shipment is still operating within acceptable service and compliance thresholds.
A shipment or an asset can look fine on a map and still be moving toward an SLA breach or regulatory issue.
That is a real gap for pharma logistics and leaves a lot of room for SLA issues.
You get visibility on the movement, but not really anything beyond that.
In the highly regulated environment of pharma logistics, the nuances of condition, handling, and the overall context of the asset determines whether a shipment is merely in transit or becoming a risk.
Teams need to know what the product has been exposed to, how long it has remained at each node, whether anything unusual happened along the route, and whether there is a complete history of movement and custody that can stand up to operational or compliance review.
Without that level of insight, tracking is of only partial value for pharma logistics. It helps explain what happened later but does very little to help teams intervene while there is still time to protect the shipment, the SLA, and the compliance record.
How AI and automated asset tracking work together to reduce SLA risk?
So by now, you know that fixing the SLA issues in pharma logistics requires more than better tracking.
It requires a system that can continuously capture operational signals and interpret them early enough to support action. This is where AI and automated asset tracking work together well.
Automated asset tracking creates a stream of usable operational data across movement, condition, and handling. That includes location across storage and transit, temperature and environmental conditions, dwell time, handoff events, and route deviations. On its own, this gives teams a stronger factual record.
With AI layered on top, that record becomes operationally useful.
AI helps identify patterns across shipments, assets, partners, and lanes. It can detect anomalies that do not match expected movement behavior, flag emerging SLA risk, and surface which exceptions need attention first.
An asset sitting longer than expected at a hub, a temperature pattern trending toward threshold breach, or a route deviation that makes an SLA miss increasingly likely are all examples of signals that matter before the final failure occurs.
AI helps connect those signals and interpret them in context. So your team goes from running an operating model pure on reporting to one based on prediction and intervention.
In pharma logistics, this matters on two levels.
- It improves service performance by helping teams respond earlier, and
- It strengthens compliance by ensuring that movement, condition, and handling are continuously recorded in a usable, time-stamped way.
That means the same system that helps prevent an SLA breach also builds a stronger audit trail for regulated movement. The point is not simply to collect more data. The point is to detect risk while there is still time to act on it.
What are some practical AI use cases in pharma logistics?
AI becomes valuable in pharma logistics when it supports concrete operational decisions.
AI doesn’t create value by adding another layer of dashboards or abstract analytics.
The real value comes from helping teams identify problems before they become bigger and impact your business severely. In that sense, the strongest use cases are the ones closest to day-to-day logistics performance.
For example, AI can help predict temperature excursion risk by analyzing live and historical condition patterns and identifying assets that are moving toward unsafe thresholds.
It can flag likely SLA delays by combining dwell time, route progression, and past performance across similar lanes.
It can identify recurring failures tied to specific routes, partners, or transfer points, making it easier to fix structural issues rather than react to isolated incidents.
It can also detect non-compliant dwell patterns, where assets remain too long at a node and create risk for both delivery commitments and regulatory expectations.
Just as importantly, AI can help teams prioritize recovery actions by identifying which assets represent the highest operational or compliance risk, instead of forcing equal attention across every alert.
These examples highlight how AI helps while the shipment, asset, or condition issue is still manageable.
The reaction time and how early you detect and manage an issue is crucial for pharma logistics SLAs.
Moving reactive SLA management to predictive control
Most current pharma logistics environments are built to react to the issues that already occurred.
That’s no longer enough. In a supply chain where service levels, product condition, and compliance requirements are tightly connected, teams need earlier signals and better operational context.
With SensaTrak’s, AI-powered analytics platform, pharma logistics operations better adapt to asset handling challenges and SLA fixes.
We help pharma logistics operators move from delayed alerts to early signals, from fragmented records to much better context, and from firefighting SLA failures to proactively reducing them.